
BIGBEAR
PHARMACEUTICAL
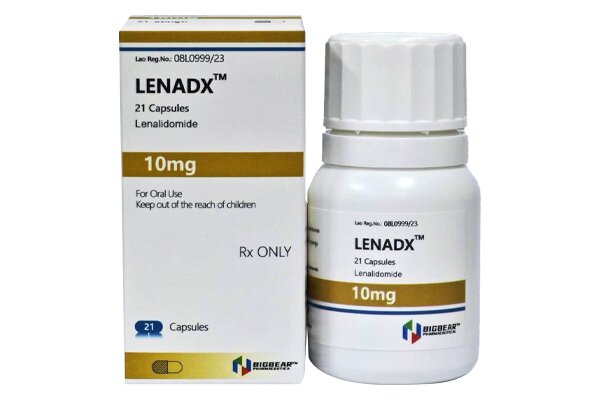
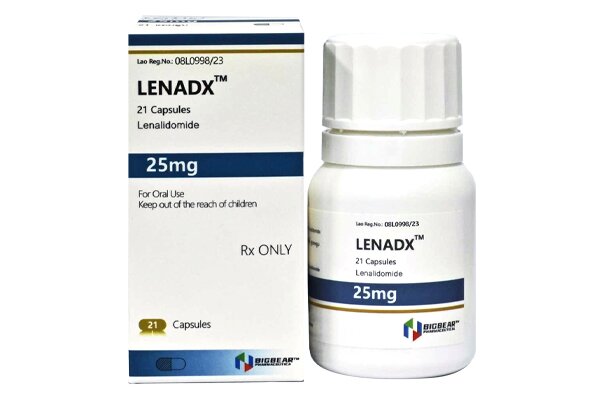
Lenalidomide is an immunomodulatory agent and a thalidomide analogue with anti-angiogenic and anti-tumor properties.

 Authentic
Authentic Guarantee
Guarantee Fast Delivery
Fast Delivery Privacy
Privacy Lenalidomide is mechanism of action mainly involves targeted binding to Cereblon (CRBN), regulating the activity of the E3 ubiquitin ligase complex, and promoting the ubiquitination and degradation of specific substrate proteins (such as Ikaros, Aiolos, and CK1α), thereby inhibiting tumor cell proliferation and inducing apoptosis.
This product is primarily indicated for the treatment of multiple myeloma, a malignant tumor affecting plasma cells in the bone marrow.
Based on the patient’s treatment phase and physical condition, this product is mainly indicated for the following three patient populations:
1.In combination with dexamethasone, for adult patients with newly diagnosed multiple myeloma who are not eligible for hematopoietic stem cell transplantation.
2.In combination with dexamethasone, for adult patients with relapsed multiple myeloma who have received at least one prior therapy (e.g., chemotherapy or targeted therapy).
3.In combination with rituximab, for adult patients with previously treated follicular lymphoma who require continued therapy (note: the lymphoma grade must be within the 1–3a range).
Special attention is required: Effective contraception must be consistently used for 4 weeks before starting treatment, throughout the entire treatment period, during any treatment interruptions, and for at least 4 weeks after treatment completion. Even if a woman of childbearing potential experiences amenorrhea, she must strictly adhere to all contraceptive requirements.
Treatment must be fully supervised by a physician with experience in managing multiple myeloma. Do not initiate treatment if the absolute neutrophil count (ANC, a key indicator of infection resistance) is < 1.0×10⁹/L, or the platelet count (related to hemostatic function) is < 50×10⁹/L.
Take at approximately the same time each day (e.g., fixed time in the morning or after dinner).
Swallow the capsule whole; do not open, crush, or chew it.
Take with a sufficient amount of warm water; may be taken on an empty stomach or with food.
Initial dose for adults: 25 mg once daily.
28-day treatment cycle: Administer daily for the first 21 days (Days 1–21), followed by 7 days of rest.
Concomitant dexamethasone: 40 mg orally on Days 1, 8, 15, and 22 of each 28-day cycle.
Continue treatment until disease progression.
Package insert content may vary by manufacturer (e.g., specific precautions or storage conditions). If there is a discrepancy between the package insert and the information provided by your physician, contact your treating physician or pharmacist immediately for confirmation.
This medication should be taken at a fixed time each day.
1.If the dose is missed by less than 12 hours, take the missed dose immediately.
2.If the dose is missed by more than 12 hours, skip the missed dose and resume the regular schedule the next day.
Important Note: Under no circumstances should a double dose be taken to make up for a missed dose.
Do not donate blood during treatment or within one week after discontinuation. Monitor for thromboembolic symptoms closely; seek immediate medical attention if symptoms such as sudden shortness of breath, chest pain, or unilateral swelling of the arm or leg occur.
1.It is unknown whether the drug is excreted in breast milk; breastfeeding is recommended to be suspended during treatment.Pregnant women, women planning pregnancy, and pediatric patients.
2.Adverse reactions such as fatigue, dizziness, drowsiness, and blurred vision may occur, which could affect the safety of driving or operating machinery. Carefully assess physical condition before engaging in such activities.
3.Elderly patients are more prone to renal function decline; pay special attention to dose adjustment and regular renal function monitoring during treatment.Breastfeeding women, elderly patients, and patients with renal impairment.
his medication may affect multiple body systems:
Cardiovascular system: Possible venous thrombosis in the extremities, swelling of hands and feet.
Metabolic system: Possible elevated blood glucose, decreased blood potassium/magnesium, hypothyroidism.
Respiratory system: May cause cough, dyspnea, runny nose, epistaxis, pneumonia, or pulmonary embolism (pulmonary vascular occlusion).
Musculoskeletal system: Possible joint pain, back pain, muscle aches, muscle weakness.
Urinary system: Possible dysuria, urinary tract infection, renal issues.
Nervous system: May cause headache, dizziness, sleep disturbances, numbness or tremors in hands and feet.
Psychiatric status: Possible fatigue and weakness.
Gastrointestinal system: Common nausea, abdominal pain, constipation, diarrhea; may be accompanied by abnormal liver function.
Hematologic system: Possible anemia, leukopenia, thrombocytopenia.
Skin reactions: Common dry skin, pruritus, rash, urticaria.
The following groups are strictly prohibited from using lenalidomide:
1.Pregnant women.
2.Women of childbearing potential who do not fully meet contraceptive requirements.
3.Patients with hypersensitivity to the active ingredient or any excipient.
4.Patients under 18 years of age (no clinical data available for this population).
Report any discomfort to your physician promptly during treatment; the physician will decide whether to discontinue the drug based on the specific situation. Pay special attention to the following:
1.Patients with a history of severe rash due to prior thalidomide use are contraindicated for this medication.
2.Consider discontinuation if a significant rash (impacting daily life) occurs.
3.Discontinue immediately and permanently if any of the following severe reactions occur: facial/throat swelling, widespread blistering and peeling of the skin, or life-threatening severe rash.
If any issues arise, please contact us immediately.
Email:laosbigbear@gmail.com

Copyright2024@ BIGBEAR All right reserved Bigbear | Bigbear Pharmaceutical | Bigbear Laos



